Larotrectinib Shows Significant Efficacy in NTRK+ Salivary Gland Tumors

Treatment with larotrectinib (Vitrakvi) in patients with various subtypes of salivary gland tumors led to a high rate early and durable responses, according to a presentation during the American Head & Neck Society 10th International Conference on Head and Neck Cancer.1
“Larotrectinib demonstrated robust and durable efficacy in patients with NTRK fusion salivary gland tumors with different types of histology,” said Xiuning Le, MD, PhD, assistant professor in the Department of Thoracic/Head and Neck Medical Oncology at The University of Texas MD Anderson Cancer Center in Houston. “These data highlight the clinical importance of identifying NTRK gene fusions in patients with salivary gland tumors, regardless of histology.”
NTRK gene fusions occur in an estimated 5% of cases of salivary gland tumors, including in approximately 80% of secretory carcinomas.
Larotrectinib was approved for the treatment of adult and pediatric patients with solid tumors harboring NTRK gene fusions, regardless of tumor type, without a known acquired resistance mutation that are either metastatic or where surgery is not recommended.2 Across a number of clinical trials, the TRK inhibitor induced an objective response rate (ORR) of 75% (95% CI, 61%-85%) in 206 adult and pediatric patients with TRK fusion–positive cancer.
For the analysis of patients with TRK fusion–positive salivary gland tumors, investigators looked at patients from the phase 2 NAVIGATE basket trial (NCT02576431) and a phase 1 trial (NCT02122913) of adult patients, all with advanced solid tumors. Twenty-four patients were treated with 100 mg larotrectinib twice daily, except for 1 patient who received 150 mg twice daily.
The primary end point was ORR by investigator assessment per RECIST v1.1 criteria, and secondary end points included duration of response (DOR), progression-free survival (PFS), overall survival (OS), and safety.
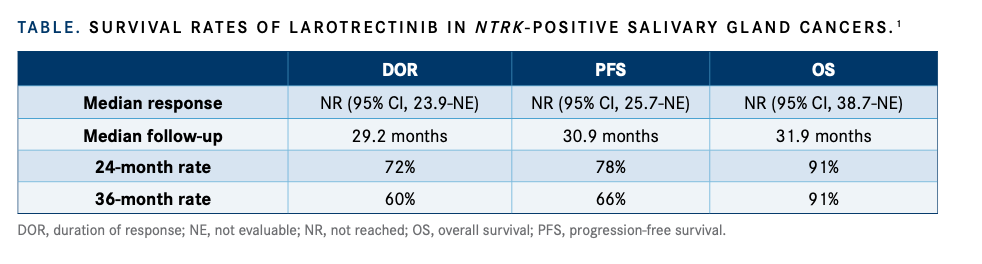
TABLE. SURVIVAL RATES OF LAROTRECTINIB IN NTRK-POSITIVE SALIVARY GLAND CANCERS.
Patients had a median age of 58.5 years (range, 28.0-78.0), 75% were male, and half had an ECOG performance status of 0. Almost all patients had undergone surgery (96%) and radiotherapy (83%), and half had received systemic therapy. Twenty-one percent of patients had received 2 or more prior systemic therapies.
Tumor histology was secretory carcinoma in 54%, adenocarcinoma in 21%, and mucoepidermoid in 13%. Also, 1 patient each had adenoid cystic carcinoma, glandular sarcomatoid carcinoma, and not otherwise specified. All patients had an ETV6-NTR K3 gene fusion.
The ORR was 92% (95% CI, 73%-99%), which consisted of complete responses in 13% and partial responses in 79%; progressive disease was reported in the other 8% of patients, 1 of whom had an NTRK3 G623R resistance mutation after prior entrectinib (Rozlytrek) therapy. Responses were observed in all histologies.
The duration of treatment ranged from 0.95 to 60.4+ months. Five of 8 patients who progressed continued treatment beyond progression for 2.8 to 20.5+ months. The median time to response was 1.84 months (range, 0.99-5.98). Le noted that early responses were “consistent with a potent, efficacious, on-target [tyrosine kinase inhibitor].”
The median DOR, PFS, and OS were all not reached as of data cutoff (TABLE 1). The DOR rate was 72% at 24 months and 60% at 36 months. The PFS rate was 78% at 24 months and 66% at 36 months. The OS rate was 91% at both 24 and 36 months.
Le noted that the treatment was well-tol-erated overall, even for long-term use. Treatment-emergent adverse events (AEs) were mostly grade 1 or 2. No grade 4 events or treatment discontinuations due to treatment-related AEs were reported in the study.
The most common treatment-related AEs were alanine aminotransferase (ALT) increase, dizziness, aspartate aminotransferase (AST) increase, fatigue, constipation, nausea, myalgia, and muscle spasm. Grade 3 treatment-related AEs included ALT increase in 3 patients, AST increase in 2, dizziness in 1, and abnormal hepatic function in 1.
REFERENCES
1. Le X, Baik C, Bauman J, et al. Efficacy and safety of larotrectinib in patients with advanced TRK fusion salivary gland cancer. Presented at: American Head & Neck Society 10th International Conference on Head and Neck Cancer; July 22-25, 2021; virtual. Abstract S030.
2. FDA approves larotrectinib for solid tumors with NTRK gene fu-sions. FDA. Updated December 14, 2018. Accessed August 24, 2021.
link








