Malaria prevention in children and pregnant women: a review of gaps and emerging challenges in sub-Saharan Africa
Despite ongoing global efforts to reduce disease burden, malaria incidence rose to 60.4 cases per 1000 population at-risk in 2023, from 58.6 in 2022. This translated to 597,000 deaths globally, with children under five years accounting for approximately 20% of mortality1. Pregnant women and children under the age of five remain the most vulnerable populations in malaria-endemic countries2. This concerning trend underscores the urgent need to optimize life-saving interventions to address remaining gaps in malaria prevention strategy implementation. Malaria remains a public health challenge, particularly in low and middle-income countries (LMICs), with a disproportionate burden in sub-Saharan Africa (SSA)3,4. The World Health Organization (WHO) recommends a package of interventions for these high-risk groups; including intermittent preventive treatment in pregnancy using sulfadoxine-pyrimethamine (IPTp-SP) and insecticide-treated nets (ITNs) for pregnant women, and prompt diagnosis and treatment, insecticide-treated nets ITNs, and more recently, malaria vaccines for children. Integrated interventions have averted significant malaria disease burden, reducing global mortality and incidence by 32.6% and 21.1%, respectively between 2000 and 20212, underscoring the importance of comprehensive harmonized approaches to sustainably control malaria.
However, implementation gaps still persist in spite of significant investment in malaria control programs. In 2023, out of approximately 36 million pregnancies, 12.4 million (34%) were infected with malaria in moderate and high malaria endemic countries in sub-Saharan Africa1. Contracting malaria during pregnancy bears substantial risks to pregnant women and girls, the fetus, and new-born child possibly leading to severe disease and mortality. These risks include: accumulation of parasite infected red-blood cells within the placenta leading to maternal anemia, elevating the risk of maternal mortality and increasing the risk of stillbirths, preterm births, and neonatal mortality1. Placental infection consequently leads to poor fetal development and low-birthweight associated with delayed growth, poor cognitive outcomes1,5,6,7, and correlates with adult disease8. This implies that coverage of essential interventions remains suboptimal among vulnerable populations, with inequities in access and utilization. Health equity and inclusion that ensure equitable access to life-saving interventions that include: rapid diagnostic testing, vaccines, drugs, treated bed-nets, and early diagnosis at health facilities are important to accelerate progress toward malaria elimination. Continuous evaluation to understand gaps is crucial for developing targeted strategies to reach the most vulnerable populations. Meta-analyses of factors influencing delivery, access and use of malaria control and prevention interventions highlight socioeconomic status, educational attainment, number of antenatal care (ANC) visits, and frail health systems as weak points2. Here, we review recent evidence on the implementation of IPTp-SP, ITNs, and early fever diagnosis at health facilities as malaria prevention strategies for pregnant women and children under five in sub-Saharan Africa that remain below the WHO global 2030 malaria reduction targets (Fig. 1, Supplementary Data). We evaluate the implementation gaps and emerging challenges to identify key socioeconomic determinants of intervention uptake and propose context-specific solutions to accelerate progress toward elimination targets. To guide this narrative review, we conducted a targeted literature search on PubMed and Google Scholar using the terms ‘malaria’, ‘Africa’, ‘sub-Saharan Africa’, ‘maternal’, ‘child’, ‘children’, ‘IPTp-SP’, ‘ITNs’, ‘malaria diagnosis and treatment’, and ‘malaria vaccines’. Articles were included based on their relevance to maternal and child malaria prevention, diagnosis, or treatment in sub-Saharan Africa. Additional references were identified through citation tracking.
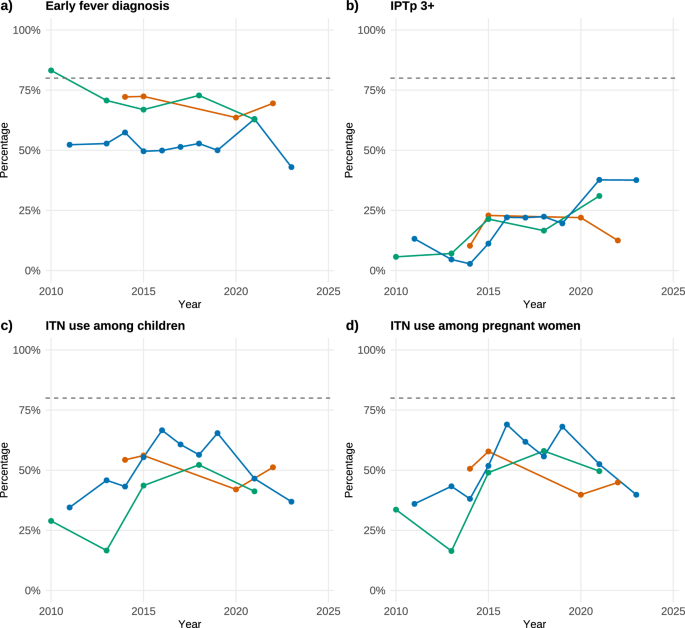
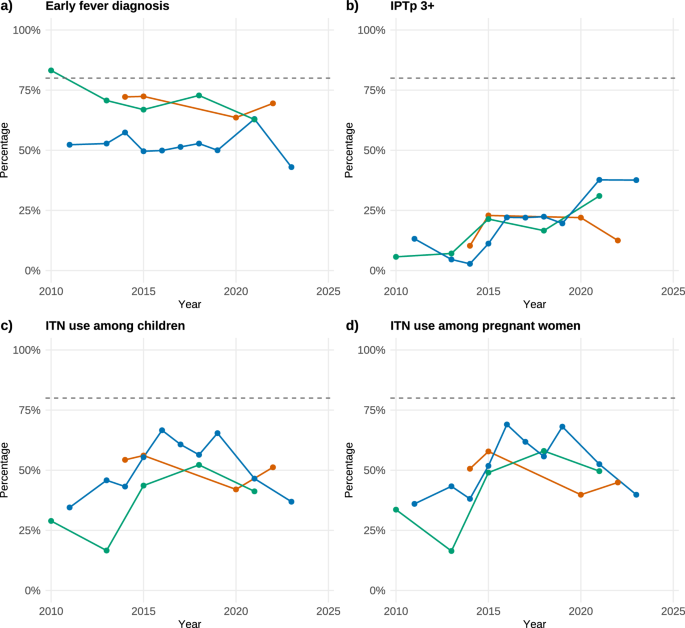
a Care-seeking for fever among children under five at a health facility, representing early diagnosis for suspected malaria. b Coverage of intermittent preventive treatment in pregnancy with sulfadoxine–pyrimethamine (IPTp-SP), shown as the proportion of pregnant women who received at least three doses (IPTp-3 + ). c Use of insecticide-treated nets (ITNs) among children. d Use of insecticide-treated nets (ITNs) among pregnant women. The horizontal dashed line indicates the World Health Organization (WHO) 2030 target of achieving ≥80% coverage for key malaria prevention and case-management interventions. Data are derived from Kenya, Senegal, and Nigeria and were selected based on availability the indicators in national demographic and health surveys conducted between 2010 and 2022. All three countries demonstrate substantial gaps between current coverage levels and the 2030 targets. Publicly available demographic health survey (DHS) and malaria indicator survey (MIS) data were retrieved from The DHS Program STATcompiler ( which consists of demographic and health indicators.
Progress made
In the past two decades considerable efforts have been made towards global elimination of malaria as a public health threat. The RTS,S (Repetitive, T cell epitope, Surface antigen, Surface antigen) vaccine pilot was successful following trials in Kenya, Ghana and Malawi paving way for inclusion into routine childhood immunization programs9,10,11,12. This complements the more recently approved, cheaper and easier to produce R21 vaccine, also administered in childhood13. The widespread deployment of artemisinin-based combination therapies (ACT) for treating uncomplicated malaria has been effective, particularly for Plasmodium falciparum malaria as an alternative to mono-therapies, and has steadily led to a decline in malaria in African and Asian countries14. However, resistance to ACT has emerged and become localised in Southeast Asia, with recent reports of emergence and spread of artemisinin resistance in East Africa were reported in Uganda and Rwanda15,16. A shift in partner drugs used against parasites restricts wide-spread multi-drug resistance by imposing a fitness cost to the parasite17. This strategy has been recommended by WHO for first-line antimalarial treatment when cure efficacy falls below 90%15,18, and different ACTs have been proposed for use as first-line treatment, either sequentially or simultaneously19,20,21. Robust molecular surveillance of circulating parasites is essential to monitor and inform emerging ACT non-susceptibility potentially delaying drug resistance21,22,23,24.
The malaria control landscape has witnessed a strategic shift from conventional pyrethroid-only insecticide-treated nets (ITNs), long considered the cornerstone of prevention efforts, toward “next-generation bed nets”, for areas with documented high pyrethroid resistance25. These next generation nets provide complementary modes of action to pyrethroids, restoring susceptibility in resistant mosquitoes. An example is PBO (piperonyl butoxide) a chemical that enhances the potency of pyrethroids against resistant mosquitoes by inhibiting cytochrome P450s (P450s) from detoxifying pyrethroids25. Overall, next generation bed nets have led to a general reduction in malaria transmission in East and West Africa and outperformed pyrethroid-only insecticide-treated nets25. However, the distribution of these limited number of nets in endemic areas is mostly done during eradication campaigns resulting in low coverage coupled with the short lifespan of the insecticide active-ingredients in spite of increasing populations of pyrethroid-resistant mosquitoes26.
Early predictions on the effect of gradual global temperature increases on malaria transmission showed slight temperature increases resulted in substantial risk of transmission27. This places malaria non-endemic areas, and especially those in countries with fragile healthcare systems, at risk of future outbreaks28 due to environmental changes that favor expansion of vectors and increased transmission of disease29. Evidence suggests these climatic changes might become more suitable for malaria transmission in tropical highland regions, which typically have minimal malaria incidence30. Climatic impact assessment is therefore important when developing detailed maps for malaria vector control and transmission measures in addition to socioeconomic and structural factors31,32. The effects and magnitude of climate change on malaria and other diseases is likely to vary by geography and over time within endemic regions, with women, children, and the elderly being most at-risk of morbidity associated with these changes33. Engaging local communities in malaria endemic regions on the connection between malaria prevalence and climate change is therefore crucial34. Prioritizing strategies to manage the effects of climate change on malaria eradication policies in Africa will ensure the success of prevention and control programs35,36.
There has been a decline in malaria deaths in children under the age of 5 years from 87% to 74% between 2000 and 20231. Licensure of R21 and RTS,S vaccines, which are high impact tools for prevention of malaria among children, and their scale-up through inclusion in routine childhood immunization programs has already increased child survival and health by up to 75%1,11. Intermittent preventive treatment in pregnancy (IPTp) using sulfadoxine-pyrimethamine (SP), regardless of parasitemia or symptoms, is recommended to reduce malaria incidence among pregnant women. However, low uptake in SSA suggests gaps in access and uptake of this intervention by this at-risk group37,38. There has been an expansion of IPTp alongside antenatal clinic visits and child vaccination programs, and increased uptake has been associated with reduced maternal deaths39,40. Despite this progress the Global Technical Strategy (GTS) target of 90% reduction of malaria cases and deaths by 2030 is likely to be unmet, since the 2023 global malaria incidence is nearly three times higher than required to reach the target.
Prevention strategies and challenges intermittent preventive treatment (IPTp-SP)
Malaria disproportionately affects women in endemic regions, compounding the health risks of pregnancy and reinforcing cycles of inequality. The revised 2016 WHO ANC guidelines that expanded the number of third trimester visits from two to five, aims to reduce preventable maternal and fetal morbidity by promoting early detection and timely management of complications, including malaria41. WHO recommends intermittent preventive treatment with sulfadoxine–pyrimethamine (IPTp-SP) for all pregnant women living in areas of moderate-to-high malaria transmission. Administration should begin as early as possible in the second trimester and continue at monthly intervals until delivery2. Despite this guidance, IPTp-SP coverage across sub-Saharan Africa remains inadequate, with most countries achieving less than 60% coverage for the recommended three-dose regimen (IPTp3 + ), well below the WHO recommended 80% national target6,42,43,44,45,46,47. Suboptimal coverage reflects a complex interplay of individual, health system, and structural barriers. Timely and frequent antenatal care (ANC) attendance is essential for IPTp-SP delivery. The WHO’s 2016 ANC model recommends eight contacts during pregnancy, offering up to six opportunities for IPTp-SP administration41. However, delayed initiation of ANC and irregular attendance remain widespread, undermining the potential to achieve full coverage. Health system deficiencies exacerbate the problem. Frequent stock-outs of SP are reported in several endemic countries, including Kenya, Cameroon, Ghana, and Tanzania, and inadequate provider training and inconsistent adherence to national guidelines result in missed opportunities to deliver IPTp-SP during ANC visits5,38,45,48,49. At individual-level, knowledge and awareness are critical factors that determine uptake, and women who are informed about the protective benefits of IPTp-SP are more likely to complete the recommended doses, and vice-versa for limited understanding of the dosing schedule and concerns about side effects5. Education and empowerment are pivotal, as women with tertiary education and greater autonomy in health-related decisions are significantly more likely to receive at least three IPTp-SP doses, highlighting the need for interventions that address gender and educational inequities6. Community engagement involving mentor mothers and male partners and community-based delivery of IPTp-SP promises to expand access, particularly for women who face logistical or financial barriers to ANC attendance, and increase IPTp-SP coverage5,50. However, its success depends on resolving systemic constraints such as drug supply continuity and ensuring adequate training and compensation for community health workers42. Investing in health workforce training and retention in rural areas, ensuring consistent drug availability, improving community education, and formalizing the role of community health workers in IPTp-SP delivery will address these gaps. In addition, integration of maternal and child health services with routine immunization in low- and middle-income countries through strategies like co-location, expanded staff responsibilities, and enhanced information provision improves maternal and child health outcomes while benefiting or maintaining immunization coverage39.
Insecticide-treated nets (ITNs)
WHO advocates the use of insecticide-treated nets (ITNs) as a primary strategy for malaria control during pregnancy and among children. The expanded use of next-generation dual-insecticide nets has increased significantly, accounting for nearly 80% of all nets delivered in sub-Saharan Africa in 2023, up from 59% the previous year1,51. These new nets have greater impact against malaria than standard pyrethroid-only nets and help address the growing challenge of insecticide resistance1,51. However, 28 million pregnant women in sub- Saharan Africa still do not receive ITN services7. ITN use among pregnant women in East Africa remains suboptimal at approximately 52%, with significant variation between countries well below the 2030 target of 80%7. ITN use was higher among older, married, and more educated women, while large family size was negatively associated, likely due to insufficient nets or limited sleeping space51. Despite distribution campaigns, usage gaps and inequalities persist, particularly in poorer rural and underserved communities52,53. There is significant variability in ITN use and coverage within countries2. Seasonal variation in ITN use has been observed, with higher utilization during peak transmission periods that typically coincide with rainy seasons54,55. Persistent socioeconomic inequalities exist in the uptake of ITNs, with utilization trends revealing widening disparities in countries like Kenya and Rwanda2. Evidence from Kenya challenges misconceptions about subsidized products being undervalued or misused. While bed net demand decreases sharply with increased prices (falling below 20% uptake for prices above $2.50), free distribution leads to both high adoption and sustained usage, with 90% of recipients still using free bednets one year later56. This highlights the need to target and engage socioeconomically poor communities in education campaigns focused on the importance and proper ITN usage, especially rural and marginalized areas to optimize malaria prevention programs.
Prompt febrile illness diagnosis and treatment
The majority of malaria deaths occur at home without timely or appropriate medical intervention. Early symptoms are often indistinguishable from other febrile illnesses, underscoring the importance of prompt care-seeking at an appropriate health facility. Timely diagnosis and treatment, ideally within 24 h of fever onset, is critical to reducing malaria-related morbidity and mortality, particularly among children under five years57. A low rate of early and appropriate care seeking behavior for children under five years with fever has been observed in different settings in SSA57,58,59,60,61. Gender disparities exist in care-seeking patterns, with caregivers more likely to seek prompt care for male children than female children57,61. This gender bias may reflect broader sociocultural perceptions that prioritize male children as future breadwinners57. Caregiver characteristics also influence care-seeking behavior. Older caregivers (25-49 years) and those with primary education were more likely to seek prompt care compared to younger caregivers (15-24 years)57. Barriers to prompt care seeking for febrile illness in children include economic constraints, distance to health facilities, and limited caregiver knowledge, with both individual and health system factors significantly influencing care-seeking behavior62. Targeted interventions should focus on improving caregiver awareness, especially among younger caregivers, and promoting equitable, timely care for all children regardless of sex, to reduce the high burden of morbidity and mortality.
Vaccination programs and emerging challenges
Vaccination programs targeting children under 5 years now include the WHO-recommended RTS,S and R21/Matrix-M malaria vaccines, which are being integrated into routine childhood immunization programs. These vaccines complement existing prevention strategies and could save tens of thousands of young lives annually. Initiatives like the optimizing malaria vaccine (OPT-MVAC) consortium leverage partnerships between African and European research institutes to promote vaccine access and uptake by raising awareness about vaccination importance alongside other interventions63.
Climate variability is altering malaria transmission patterns, with increased risks observed in highland areas and previously low-risk zones29,30. These changing patterns necessitate climate-informed malaria surveillance and control strategies to effectively respond to shifting transmission patterns and coordinate resources appropriately.
Although children younger than 5 years and pregnant women remain most vulnerable to malaria-associated morbidity and mortality, there is growing concern about increasing disease prevalence among school-age children64,65,66. This demographic often presents asymptomatically and shows no association between malarial parasitemia and fever, despite fulfilling WHO criteria for malaria66,67. These children represent an important reservoir for human-to-mosquito parasite transmission prompting suggestions for seasonal malaria chemoprevention to reduce malaria incidence in this population66,68,69. This is because school-age children with asymptomatic malaria serve as a parasite reservoir, posing a risk of vector-borne transmission to vulnerable younger siblings (under 5 years) and pregnant mothers who are their primary caregivers. However, intermittent screening and treatment for malaria has proven ineffective in improving health outcomes or educational performance (including cognitive development and academic achievement) among school children in malaria-endemic regions70. This ineffectiveness suggests heterogeneity in parasite transmission patterns, rapid reinfection rates following treatment, variable reliability of rapid diagnostic tests, and the relatively modest contribution of malaria to anaemia etiology compared with poor nutritional status and helminth infections70,71,72. Malaria prevention interventions targeting school-age children could reduce morbidity by restricting the parasite reservoir and interrupting transmission cycles.
The emergence of artemisinin resistance in East Africa, particularly in Rwanda and Uganda, poses a significant threat to malaria control efforts. Similarly, resistance to pyrethroids in mosquito vectors has necessitated the development and deployment of “next generation” bed nets.
Africa’s health sector faces a severe financing crisis with a 70% decline in Official Development Assistance (2021–2025), coinciding with a 41% surge in public health emergencies, threatening critical malaria control programs73. Increased philanthropic support has led to a rise in investment for global malaria research; however, global challenges like the COVID-19 pandemic caused a decline in subsequent years74,75,76. Recent commitments from major donors signal a potential rebound, though funding remains below requirements for meeting global malaria goals75. For example, Kenya faces a funding gap of approximately $52 million in its malaria control efforts, impacting the procurement of essential commodities and implementation of interventions77. Despite the 2001 Abuja Declaration commitment to allocate 15% of national budgets to health, only three African countries (Rwanda, Botswana, and Cabo Verde) consistently meet this target, while most remain below 10%, a situation exacerbated by an $81 billion debt service burden projected for 202573. Structural vulnerabilities include 90% dependency on imported medical supplies and critical health worker shortages. WHO recently warned that the 2025 funding cuts could further derail progress in many endemic countries, putting millions of additional lives at risk, with over 30 African countries reporting moderate or severe disruptions to malaria services28,73,77,78. Innovative financing mechanisms and private sector partnerships are essential to bridge funding gaps and reduce dependency on external aid.
The complex interplay of these factors underscores the need for comprehensive, integrated approaches in malaria prevention programs targeting pregnant women and children. Addressing gaps identified requires targeted interventions that consider socioeconomic, cultural, and structural determinants of access and utilization to sustain and accelerate progress toward eliminating malaria.
link





